Nitrilové vyšetřovací a ochranné rukavice
Vyšetřovací a ochranné nitrilové rukavice z Nitril-butadienového kaučuku (NBR). Jednorázové rukavice bez latexu, bez urychlovačů DEHP/DOP, nesterilní, nepudrované.

Nabízené produkty
Rukavice SOFTCLINIC® mají primární využití ve zdravotnictví. Dle barev mohou být využívány ve farmaceutickém průmyslu, potravinářském průmyslu, gastronomii, kosmetickém průmyslu a dalších.
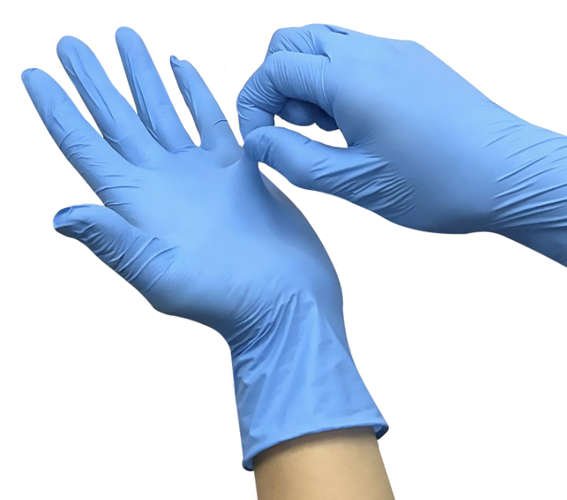
NITRILE Blue
Zdravotnictví

NITRILE Black
Zdravotnictví, Gastronomie
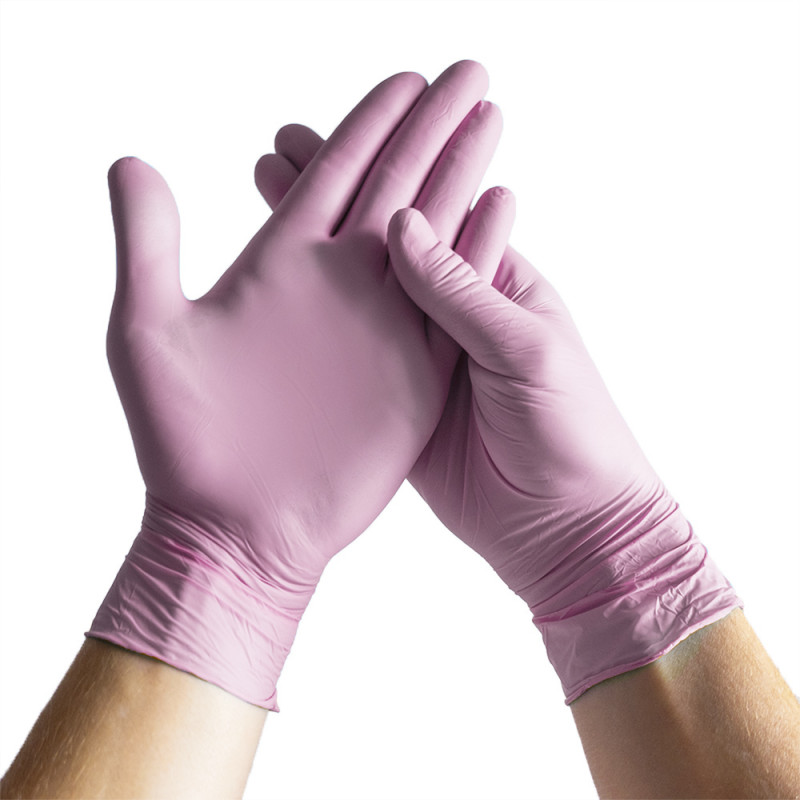
NITRILE Pink
Zdravotnictví, Gastronomie, Kosmetika
Používáno v
Velikosti
Certifikáty
Certifikáty a soubory ke stažení
Lze stáhnout produktový list, EU prohlášení o shodě nebo návod k použití.
Kde nás najdete?
Náš sortiment je aktuálně nabízen v těchto zemích:
Česká republika, Slovensko
Česká republika
Detaily produktů a balení
Kompletní informace o produktech, certifikátech, balení a oblastech využití.
-
SOFTCLINIC® rukavice jsou v souladu s nařízením EU o zdravotnických prostředcích (EU)2017/745 (zdravotní třída I.). Splňují normy EN 455-1, EN 455-2, EN 455-3, dále splňují normy EN ISO 374-1:2016 o chemických látkách a EN ISO 374-5:2016 o ochraně proti bakteriím, virům a houbám. Zároveň jsou ve shodě s EN ISO 374-4 a EN ISO 374-2 - klasifikace OOP: Kategorie III. – prostředek individuální ochrany (směrnice Rady EU 2016/425), rovněž dle norem EN 420, EN 388.
-
SOFTCLINIC® rukavice jsou používány v následujících oblastech: Farmacie, gastronomie, diagnostika, ošetřovatelská péče, laboratoře, styk s vybranými chemickými látkami, stomatologie, veterinární praxe, kosmetické salony, tetovací a piercingová studia.
-
- 1 krabička = 100 rukavic
- 1 karton = 10 krabiček = 1 000 rukavic
- 1 paleta (výška 130cm) = 60 kartonů = 60 000 rukavic
- 1 kontejner = 4.100 ks kartonů = 4.100.000 ks rukavic
Kontakt
Nenašli jste co jste hledali? Neváhejte nás kontaktovat emailem.
© 2026 Softclinic | All rights reserved








